The control of tuberculosis is an ongoing issue worldwide. A new study by Rossi and colleagues shows how genomic surveillance and a deep knowledge of the micro-scale landscape can provide invaluable insights on the effective control policies to tackle this issue.
How do novel pathogen problems emerge? Have local conditions changed, making an outbreak more likely? How did the outbreak originate, and what will the consequences be?
The resurgence in many countries of Mycobacterium bovis, the causative agent of bovine Tuberculosis in cattle, makes these questions topical. The control of bovine (or more broadly, animal) tuberculosis has proved to be very challenging with establishment in wildlife a common culprit.
M. bovis is part of the M. tuberculosis complex, and it can infect a number of mammalian hosts including badgers, and deer. While zoonotic, in developed countries this risk has been reduced by pasteurizing milk for humans’ consumption, but it remains an occupational health risk for farmers, veterinarians and slaughterhouse workers, and a serious problem for animal health and for the economy of rural areas.
As with many multi-host pathogens, the roles of different host species in bovine Tb spread are difficult to quantify, complicating the identification of good approaches to control.
Areas at high-risk for bovine TB in England are mostly in the south west and have expanded steadily for over four decades. Other parts of the country are considered low risk. Here, there are only very limited outbreaks that have been controlled by test and slaughter, with no evidence of establishment in non-cattle populations. However, in 2014 an outbreak was reported in East-Cumbria (North England), an area not contiguous to any high-risk area.
First identified in cattle, in 2017 infection was also detected in local badgers, raising concerns of incipient endemic disease. A critical initial question was whether or not the outbreak arose due to expansion via badgers, which would have implied a much greater area under threat, and potentially implying a much more rapid expansion in badgers than expected.
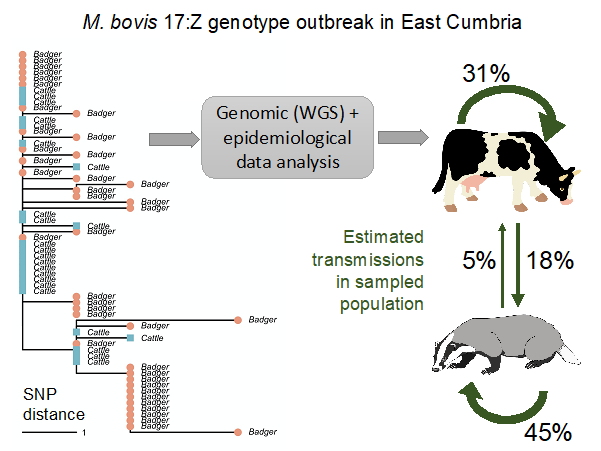
An important initial finding from our work was the identification of a close association between the genetic sequences of East Cumbria M. bovis genome samples (i.e. the bacteria DNA sequences sampled from infected hosts) with isolates taken from cattle in Northern Ireland, making introduction from cattle the mostly likely explanation.
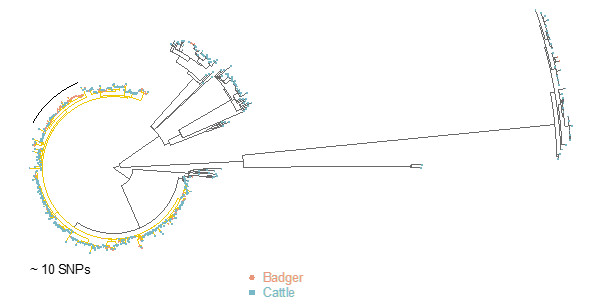
Further analyses aimed to disentangle the role of badgers and cattle across the different phases of the outbreak. While it was not possible to isolate “case 0” (i.e. the first infected animal introduced in the area), results indicated two main phases for the outbreak: initially, M. bovis circulation in the local cattle population with few potential spill-overs, followed by spread in the badger population, with badger-to-cattle transmission occurring at most infrequently.
The slow rate of establishment in badgers indicates that a more pro-active surveillance in cattle alone could mitigate the risk of insurgence in low-risk areas. If supported by biosecurity measures in farms, this could limit the spread from cattle to wildlife.
We also showed how the land parcels configuration might play a non-negligible role in the disease spread by allowing potential infectious contacts that, when considering the main farms’ building geographical location only, would be hidden. Evidence that the genetic distance between M. bovis samples was better explained when including the pattern of land parcel ownership highlights the importance of landscape characteristics in defining emergent problems.
Anecdotally, in recent years, farms decreased in number but increased in size, with individual parcels of pastureland absorbed by the remaining operations, resulting in larger farms with cattle moved between fragmented pastures. Our finding is consistent with these new land management patterns contributing to the bovine TB outbreak pattern, suggesting that, to produce effective control policies, it is necessary to tailor them to the specific ecological landscape and rearing practices, other than the cattle and wildlife populations, and their potential interplay.

These findings could have important implications for bovine TB control. Processes like climate change or reforestation can dramatically change the structure and density of wild animals’ populations, and their spatial relationships to cattle, therefore changing the risk of diseases such as bovine TB to establish in new areas. As the potential for spill-over from cattle to wildlife increases, so does the risk of new areas to become endemically infected.
While avoiding the movement of potentially infectious animals must always be a priority, policies for controlling bovine TB spread should also consider genomic surveillance, to quickly identify the origin and extent of outbreaks, as well as the risk from landscape changes at the local scale.
Our analyses suggest that in this case, cattle-related factors (movements, farm configuration) have played roles in the establishment of this outbreak. While the result itself is particular to this situation, our analysis highlights how forensic analysis of detailed genomic datasets can be crucial in understanding such outbreaks and informing their control.
Read the full paper Phylodynamic analysis of an emergent Mycobacterium bovis outbreak in an area with no previously known wildlife infections in Journal of Applied Ecology.
